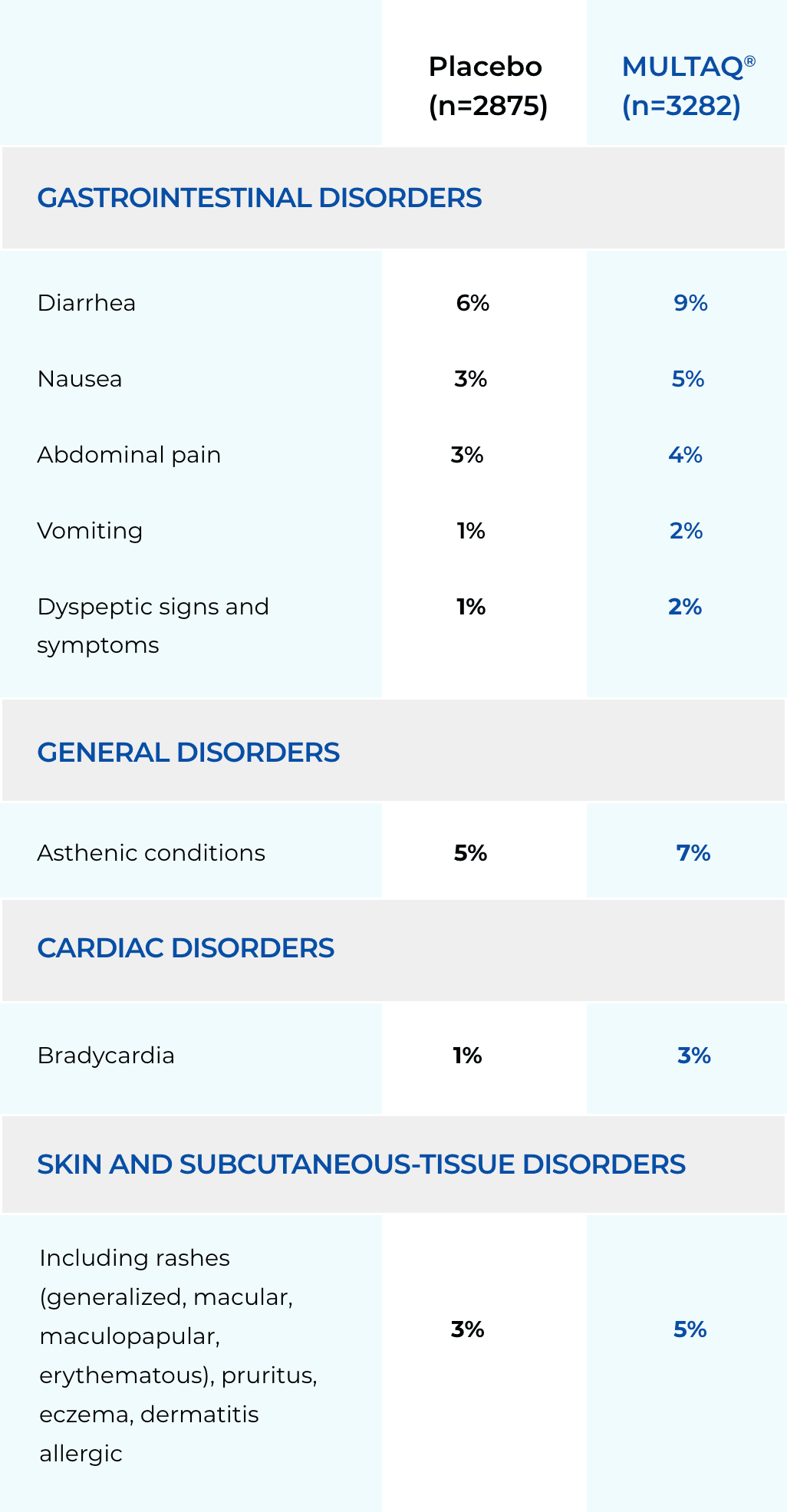
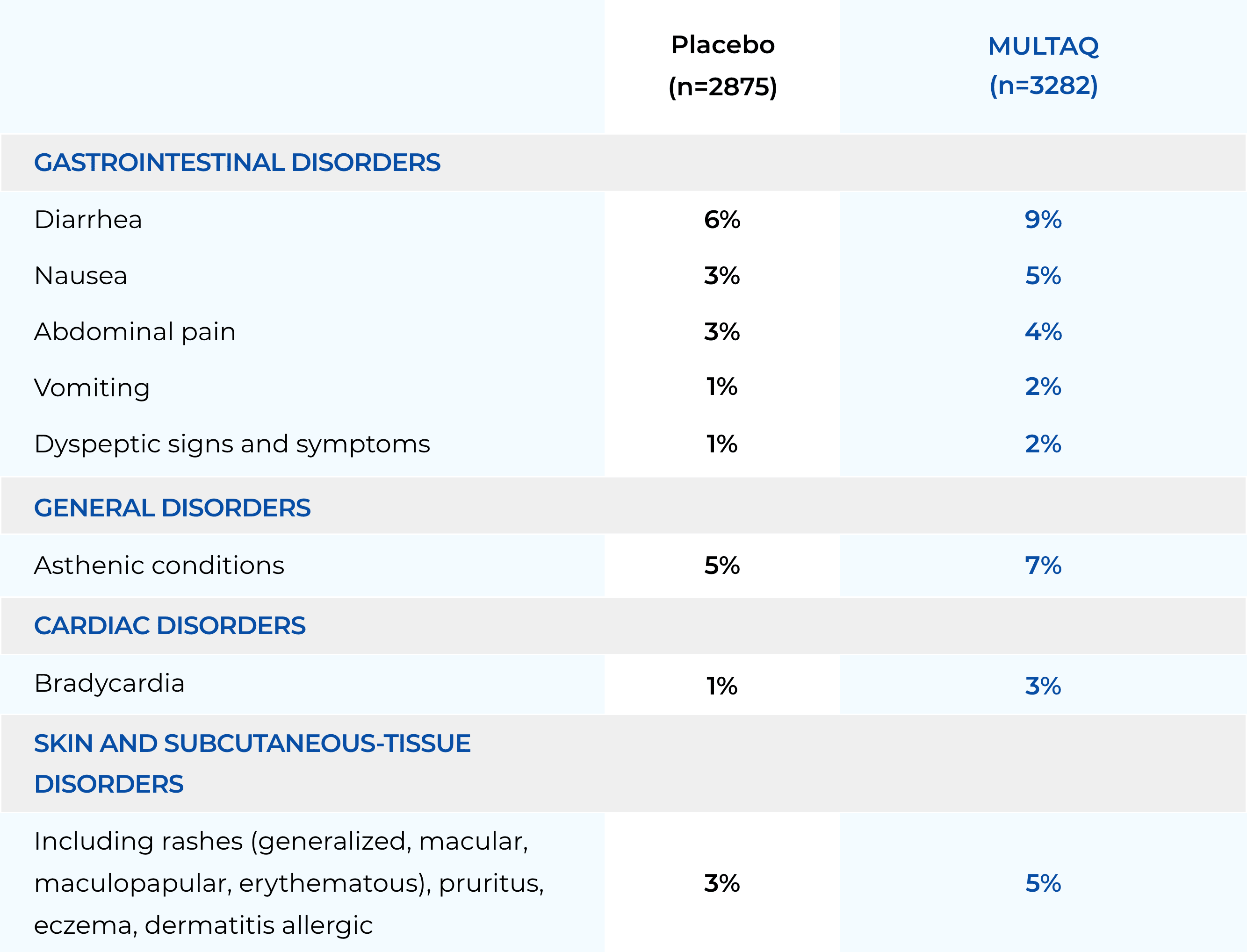
These highlights do not include all the information needed to use MULTAQ safely and effectively. See full prescribing information for MULTAQ. MULTAQ (dronedarone) tablets, for oral useInitial U.S. Approval: 2009
Blister packs of Multaq tablets. Four blister packs of Multaq tablets, two reversed. Canada Stock Photo - Alamy